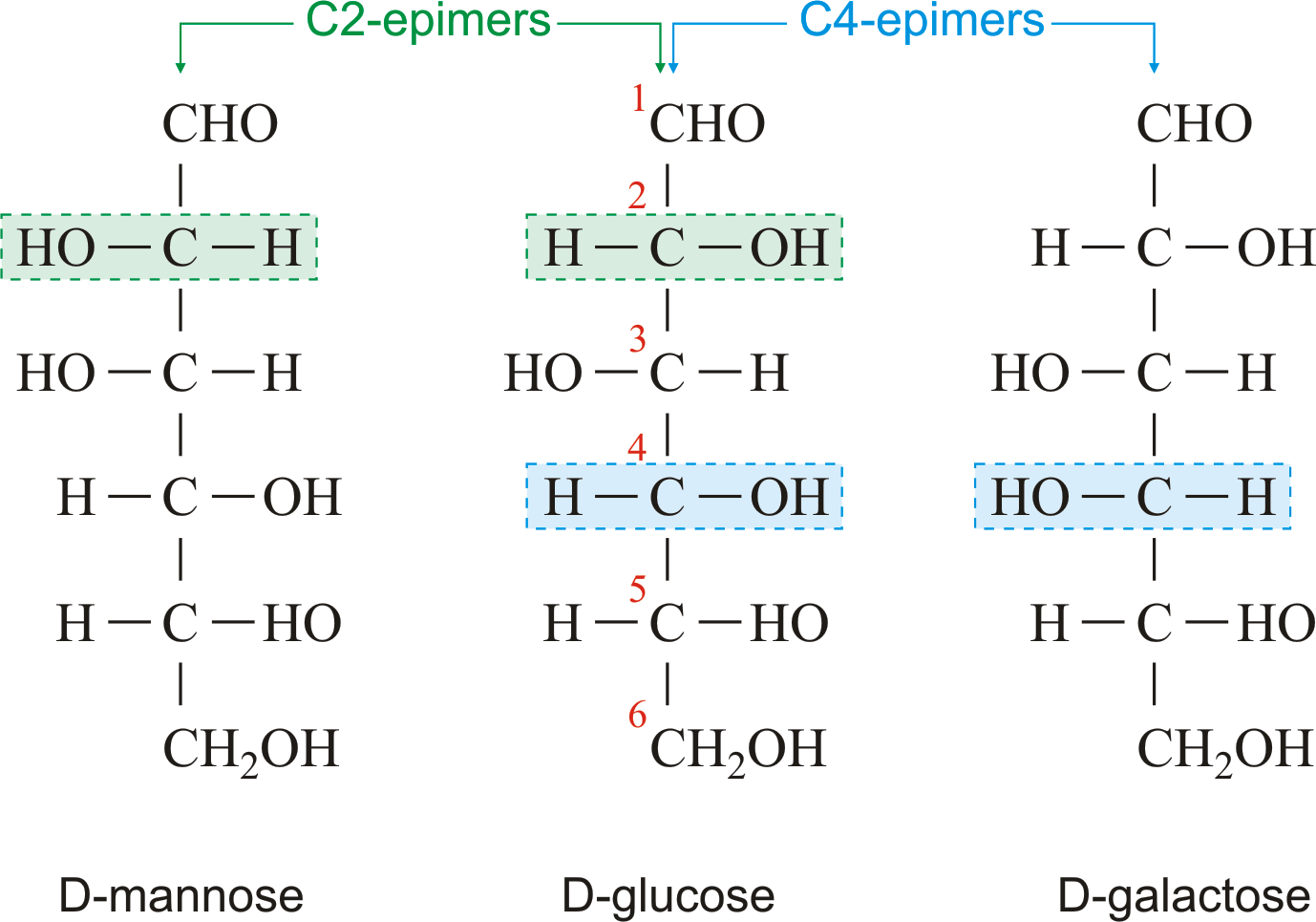
So, glyceraldehyde has two enantiomeric forms, which we call “D” and “L” glyceraldehyde.įigure 13.8 Dihydroxyacetone is not chiral The “top” and “bottom” carbon atoms are not chiral. The central carbon atom in glyceraldehyde has four different substituents, so it is chiral (Figure 13.7). Glyceraldehyde is the simplest aldose sugar. (Refer to first-year chemistry.) The “left-handed” and “right-handed” forms of a molecule are called enantiomers (mirror images).įigure 13.7 The enantiomers of glyceraldehyde are the result of the different handedness of the central carbon. This gives rise to the occurrence of optically active isomeric forms (“left-handed” or “right-handed” form of a molecule). All monosaccharides except dihydroxyacetone contain one or more chiral carbon atoms. If the four substituents on a carbon atom in a molecule are all different, then the atom and the molecule are “chiral” (asymmetric). A carbon atom with four substituents is tetrahedral, not planar. Sugar chemistry is dominated by the issue of chirality, i.e., asymmetry or “handedness” (Figure 13.6). All monosaccharides except dihydroxyacetone contain one or more chiral carbons. Monosaccharides contain asymmetric carbon atoms: ENANTIOMERSįigure 13.6 Chirality dominates sugar chemistry. He introduced much of the terminology and notation that we still use today. Fischer studied the analysis, synthesis, and stereochemistry of simple sugars. Much of our understanding of carbohydrate chemistry goes back to the work of the German chemist Emil Fischer (Figure 13.3) and his students, in the late 19 th century. Figure 13.2 shows you how the name glyceraldehyde reminds you of the three-carbon alcohol glycerol (a molecule that we have already encountered, when we discussed fats), and the name dihydroxyacetone reminds you of the three-carbon ketone acetone, which is a common organic solvent that you use in the lab.Įmil Fischer (1852-1911): Nobel Prize in Chemistry 1902 Figure 13.3 Emil Fischer There are two trioses: the aldose glyceraldehyde and the ketose dihydroxyacetone. We call these sugars “trioses” (three-carbon sugars). The simplest sugars contain three carbon atoms: one carbonyl group and two carbons bearing hydroxy groups. Glyceraldehyde is an aldose (left), while dihydroxyacetone is a ketose (right). T he simplest monosaccharides contain three carbon atoms (Trioses) Figure 13.2 Trioses are the simplest monosaccharides. The chemical properties of sugars therefore, reflect the properties of alcohols and ketones/ aldehydes – but, as we will see, they also exhibit some special properties that reflect the presence of both carbonyl and alcohol functional groups within a single molecule. Most sugars have names that end in “ose”, such as glucose and sucrose. The aldehyde-sugars are called aldoses and the ketone-sugars are called ketoses ( Figure 13.1). Therefore, sugars are polyhydroxy aldehydes or polyhydroxy ketones. Monosaccharides combine two organic chemical functional groups: they have a carbonyl (C=O) group, which is either an aldehyde (with the carbonyl carbon bonded to one carbon atom and one hydrogen atom) or a ketone (with the carbonyl carbon bonded to two other carbon atoms), and they also have at least two carbons bearing hydroxyl (alcohol) groups (-C-OH). What are sugars? Figure 13.1 Aldehyde sugars are called aldoses (left, ribose) and ketone sugars are called ketoses (right, ribulose). All monosaccharides share some chemical properties: they are very water-soluble, poorly soluble or insoluble in organic solvents such as ether or hexane, colorless, and, at least in most cases, sweet to the taste and have the approximate formula (CH 2O) n. many monosaccharide units are called disaccharides, trisaccharides … polysaccharides. Simple sugars, with only one sugar unit, are called monosaccharides. They are also found attached to many proteins (“glycoproteins”). We will be looking at the general principles of carbohydrate chemistry.Ĭarbohydrates are of great importance in energy metabolism, and are essential components of nucleic acids (RNA and DNA). However, only a few of these sugars are commonly found in cells, and for the purposes of this course, we only need to consider a few specific sugars. Unlike the proteins, which are built from a limited set of building blocks (the amino acids), the number of possible sugar molecules is very large. Sugars are the most abundant biomolecule on earth. Carbohydrates, in common speaking, are called “sugars”.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
